Download Files:
Imatinib
58 CAD – 334 CADPrice range: 58 CAD through 334 CAD
Products Details
Product Description
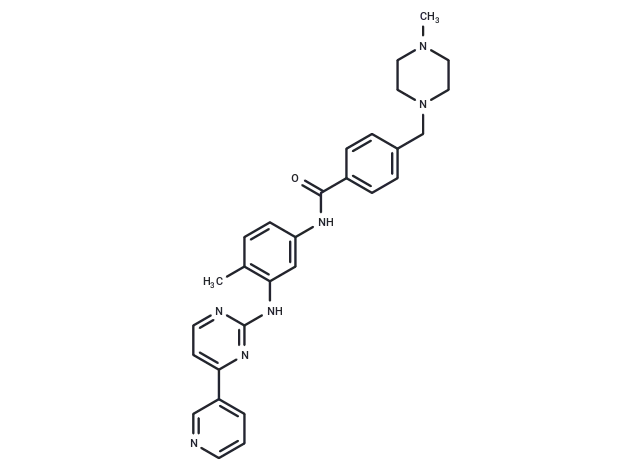
– Imatinib (STI571) is a multi-targeted receptor tyrosine kinase inhibitor that selectively inhibits the kinase activities of BCR/ABL, v-Abl, PDGFR, and c-kit with oral activity. Imatinib has antitumor activity for the treatment of chronic granulocytic leukemia.
Web ID
– T6230
Storage Temperature
– -20℃
Shipping
– Blue Ice
Molecular Formula
– C29H31N7O
Citations
– 1. Wang B, He B S, Ruan X L, et al. An integrated microfluidics platform with high-throughput single-cell cloning array and concentration gradient generator for efficient cancer drug effect screening. Military Medical Research. 2022, 9(1): 1-17.
2. Li Q, Huang Z, Peng Y, et al. RanBP3 Regulates Proliferation, Apoptosis and Chemosensitivity of Chronic Myeloid Leukemia Cells via Mediating SMAD2/3 and ERK1/2 Nuclear Transport. Frontiers in Oncology. 2021: 3349.
3. Zhou D, Yang S, Yan H, et al. SC75741, a novel c-Abl inhibitor, promotes the clearance of TDP25 aggregates via ATG5-dependent autophagy pathway. Frontiers in Pharmacology. 2021: 2891.
4. Zeng D, Gao M, Zheng R, et al. The HSP90 inhibitor KW-2478 depletes the malignancy of BCR/ABL and overcomes the imatinib-resistance caused by BCR/ABL amplification. Experimental Hematology & Oncology. 2022, 11(1): 1-14
5. Zeng D, Gao M, Zheng R, et al. The HSP90 inhibitor KW-2478 depletes the malignancy of BCR/ABL and overcomes the imatinib-resistance caused by BCR/ABL amplifcation. Experimental Hematology & Oncology. 2022, 11(1): 1-14
6. Liu H M, Guo C L, Zhang Y F, et al. Leonurine-repressed miR-18a-5p/SOCS5/JAK2/STAT3 axis activity disrupts CML malignancy. Frontiers in Pharmacology. 2021, 12.
7. Dong L, Shen S, Chen W, et al. Discovery of Novel Inhibitors Targeting Human O-GlcNAcase: Docking-Based Virtual Screening, Biological Evaluation, Structural Modification, and Molecular Dynamics Simulation. Journal of chemical information and modeling. 2019, 59(10): 4374-4382.
8. Qin R, Wang T, He W, et al.Jak2/STAT6/c-Myc pathway is vital to the pathogenicity of Philadelphia-positive acute lymphoblastic leukemia caused by P190BCR-ABL.Cell Communication and Signaling.2023, 21(1): 27.
9. He W, Liu S, Wei W, et al.mTOR inhibition by AZD2014 alleviates BCR:: ABL1 independent imatinib resistance through enhancing autophagy in CML resistant cells.American Journal of Cancer Research.2024, 14(6): 2770.
References
– Gobin B, et al. Imatinib mesylate exerts anti-proliferative effects on osteosarcoma cells and inhibits the tumour growth in immunocompetent murine models. PLoS One. 2014 Mar 5;9(3):e90795.
CAS Number
– 152459-95-5
Molecular Weight
– C29H31N7O
Compound Purity
– 0.9986
SMILES
– CN1CCN(CC2=CC=C(C=C2)C(=O)NC2=CC(NC3=NC(=CC=N3)C3=CC=CN=C3)=C(C)C=C2)CC1
Pathway
– Microbiology/Virology|||Tyrosine Kinase/Adaptors|||Angiogenesis|||Cytoskeletal Signaling|||Autophagy
Product type
– Small Compound
Disclaimer: All products are for Research use only unless clearly stated otherwise on the product datasheet. Datasheets provided on the website are drafts for reference purpose only and you are requested to always refer to the hard copy included in the kit for your experimentation. Agdia Products are available for delivery only in Canada.
Related Products
1000 in stock
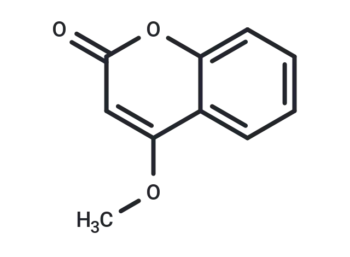
4-Methoxycoumarine
56 CAD – 266 CADPrice range: 56 CAD through 266 CAD
1000 in stock