Download Files:
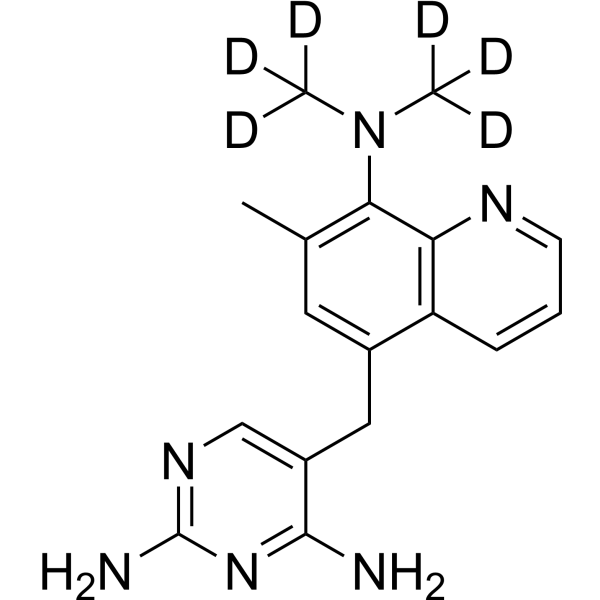
Baquiloprim-d6
SKU
HY-19581S-1mg
Category Isotope-Labeled Compounds
Tags Anti-infection;Others, Antibiotic;Bacterial;Isotope-Labeled Compounds, Infection
Products Details
Product Description
– Baquiloprim-d6 is deuterium labeled Baquiloprim. Baquiloprim, an antibiotic, is a selective inhibitor of bacterial dihydrofolate reductases. Baquiloprim possesses in vitro bacteriostatic activity against both Gram-negative and Gram-positive bacteria[1][2].
Web ID
– HY-19581S
Shipping
– Room temperature
Applications
– COVID-19-immunoregulation
Molecular Formula
– C17H14D6N6
References
– [1]Russak EM, et al. Impact of Deuterium Substitution on the Pharmacokinetics of Pharmaceuticals. Ann Pharmacother. 2019;53(2):211-216.|[2]Lewicki J, et, al. Oral bioavailability and pharmacokinetics of baquiloprim in dwarf goats. Res Vet Sci. 1995 May;58(3):268-71.|[3]White DG, et, al. Comparison of danofloxacin with baquiloprim/sulphadimidine for the treatment of experimentally induced Escherichia coli diarrhoea in calves. Vet Rec. 1998 Sep 5;143(10):273-6.
CAS Number
– 1228182-50-0
Molecular Weight
– 314.42
SMILES
– [2H]C([2H])([2H])N(C([2H])([2H])[2H])C(C(N=CC=C1)=C1C(CC2=CN=C(N)N=C2N)=C3)=C3C
Clinical Information
– No Development Reported
Research Area
– Infection
Solubility
– 10 mM in DMSO
Target
– Antibiotic;Bacterial;Isotope-Labeled Compounds
Pathway
– Anti-infection;Others
Product type
– Isotope-Labeled Compounds
Disclaimer: All products are for Research use only unless clearly stated otherwise on the product datasheet. Datasheets provided on the website are drafts for reference purpose only and you are requested to always refer to the hard copy included in the kit for your experimentation. Agdia Products are available for delivery only in Canada.